Cyclopropanes
Metal Homoenolates
Cyclopropanols readily undergo ring-opening reactions to yield metal homoenolates (Scheme 1). We reported a highly diastereoselective synthesis of cyclopropylamines from cyclopropanols (Scheme 1c). Since this initial report, we have demonstrated that readily available α-chloroaldehydes can be directly converted to cyclopropylamines via a homoenolate intermediate. We have also shown that α-tertiary cyclopropylamines can be accessed from 1- and 1,2-disubstituted cyclopropanols (i.e. via ketone homoenolates) and are currently exploring the use of nucleophiles beyond secondary amines in this chemistry.
Key References:
1) Mills, L. R.; Barrera Arbelaez, L. M.; Rousseaux, S. A. L. J. Am. Chem. Soc. 2017, 139, 11357–11360.
2) West, M. S.; Mills, L. R.; McDonald, T.; Lee, J. B.-J.; Ensan, D.; Rousseaux, S. A. L. Org. Lett. 2019, 21, 8805–8809.
3) West, M. S.; Pia, J. E.; Rousseaux, S. A. L. Org. Lett. 2022, 24, 5869–5873.
4) Scotchburn, K.; Allen, L. G.; Rousseaux, S. A. L. Org. Lett. 2026, 28, 2427-2431.
Photochemical Approaches to Cyclopropanols
Unlike cyclopropanols and cyclopropylamines, cyclopropane thiols are much more challenging to prepare, especially 2- and 2,3-substituted derivatives. Inspired by the Newman-Kwart rearrangement and our work on the use of redox-active thiocarbonyl auxiliaries (see Theme 3), we developed a photocatalytic O- to S-rearrangement of thiocarbonyl-activated cyclopropanol derivatives to access cyclopropanethiols (Scheme 2). Mechanistic studies revealed that the reaction proceeds via a radical chain mechanism, enabling us to develop a more broadly applicable strategy for the conversion of tertiary alcohols to thiols.
Key References:
1) Monteith, J. J.; Pearson, J. W.; Rousseaux, S. A. L. Angew. Chem. Int. Ed. 2024, 63.
Cyclopropanols are typically prepared via Kulinkovich or Simmons-Smith reactions. While these reactions represent powerful approaches to prepare these building blocks, they typical involve harsh reaction conditions (e.g., use of organometallic reagents) that limit functional group compatibility. Cyclobutanols can be prepared via Norrish-Yang Type II photocyclization of aryl ketones under mild conditions. However, access to 3-membered rings via this approach remains challenging due to the strong kinetic preference for 1,5- over 1,4-HAT. With this in mind, we have recently reported a strategy to access 1,3-diradicals from aryl ketones, overriding the inherent Norrish Type II reactivity of these systems. Our approach relies on the strategic incorporation of a boryl substituent at the β-carbon and the solvent controlled selective population of the π,π triplet state over the n,π triplet state, with the former preferentially undergoing 1,4-boryl group transfer over 1,5-HAT (Scheme 3).
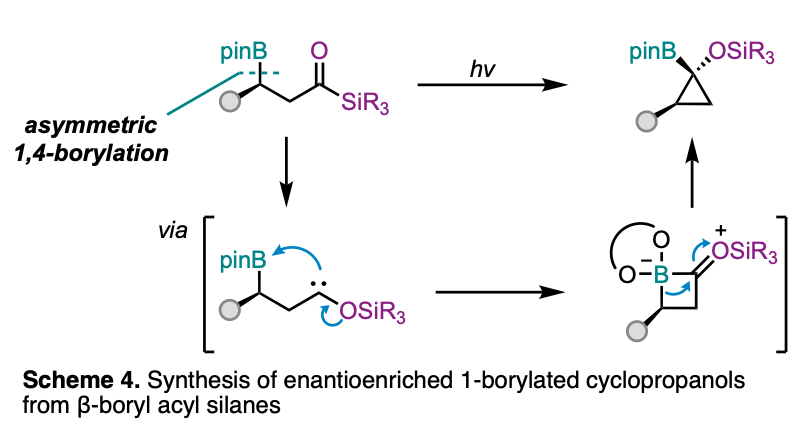
In related work, we have found that enantioenriched 1,1-siloxycyclopropylboronic esters can be synthesized from enantioenriched β-boryl acyl silanes under mild photochemical conditions with near complete (>98%) enantiospecificity (Scheme 4). Our approach hinges on the photochemical 1,2-Brook rearrangement of acyl silanes to α-siloxycarbenes, which serve as the reactive species that prompts a stereospecific 1,2-metallate rearrangement. Intriguingly, the triplet energy transfer reaction conditions to promote α-siloxycarbene formation were observed to be stereoablative, due to an unexpected secondary energy transfer pathway that led to cyclopropane racemization. Instead, direct photoexcitation of the acyl silanes enabled the cyclization with near complete stereospecificity.
Key References:
1) Pearson, J. W.; Dudra, S. L.; Palermo, A. F.; Chiu, B. S. Y.; Dang, J.; Gabbey, A. L.; Henson, B. A. E.; Hou, T. R.; Nabavi, N.; Rousseaux, S. A. L. J. Am. Chem. Soc. 2025, 147, 36890–36897.
2) Palermo, A. F.; Imbriaco, B.; Chan, S. C.; Doan, B. A.; Rousseaux, S. A. L. Synlett. 2024, 35, 895–898.
3) Palermo, A. F.; Doan, B. A.; Rousseaux, S. A. L. ACS Catal. 2025, 15, 19633–19641.